Safety- and performance studies
MDC provides support with microbiological expertise in risk-based test designs, ensures a smooth testing process and provides professional support in the evaluation and documentation of results.
in vitro efficacy studies
Depending on the intended purpose (e.g. disinfection of surfaces, hands or instruments, pre-disinfection or final disinfection), the efficacy against various germs must be proven using standardized norm test methods.
MDC helps to test time- and cost-efficiently. A test plan is drawn up according to the active ingredient tolerance of the germs, MDC organizes the testing processes, and checks and evaluates the laboratory results.
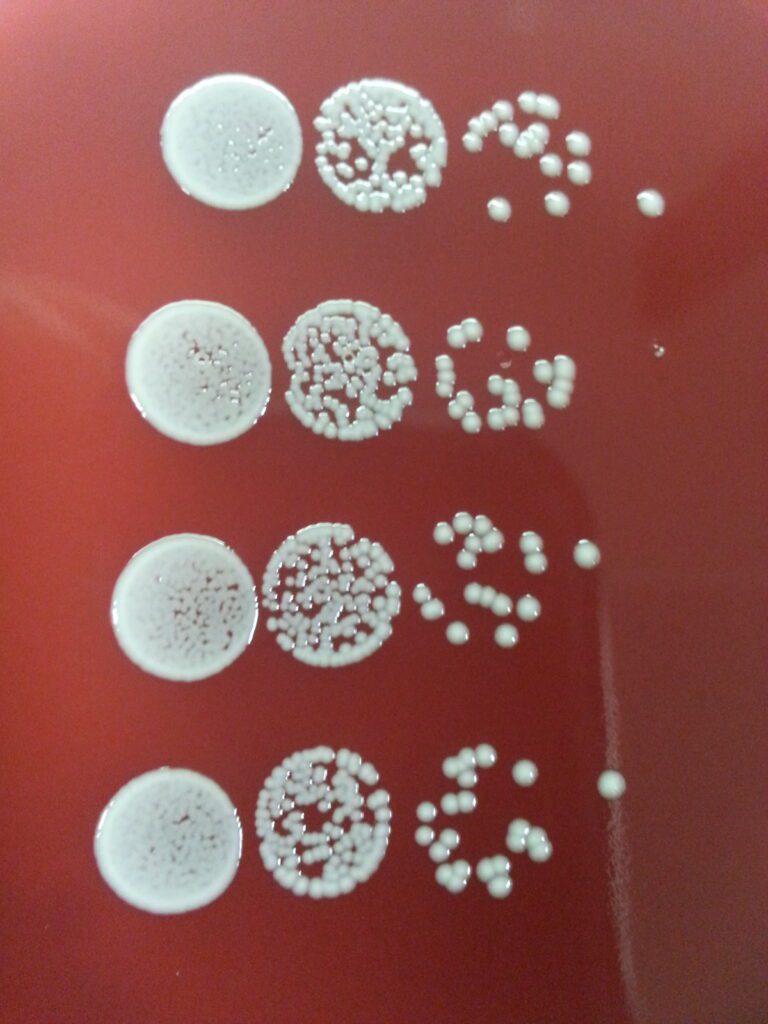

Efficacy of antimicrobial materials
Currently, the choice of standard test methods for testing antimicrobial materials is limited. MDC supports you in defining the test setup and regarding test method, organization, evaluation and documentation of the test.
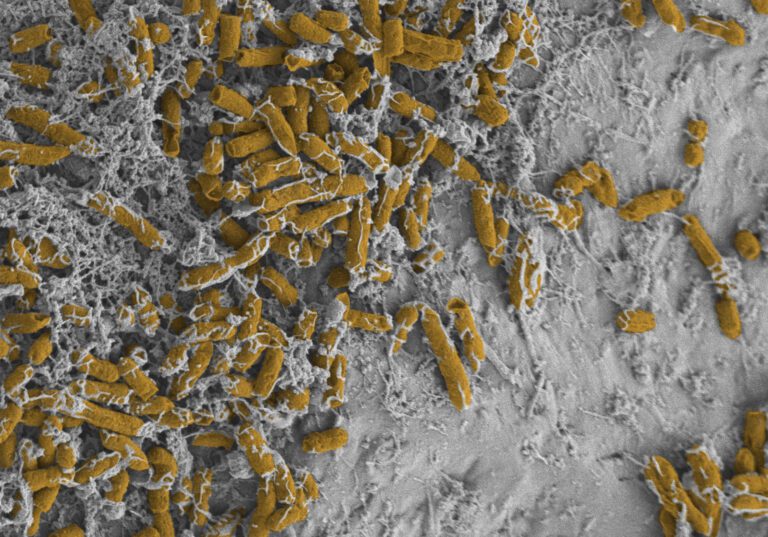
Efficacy against biofilms
In biofilms, bacteria can be up to 1000 times more tolerant to antibiotics and biocides. Currently, there is no standardized norm test method for efficacy testing against biofilms. MDC assists in the selection of the test methodology and organization of the test.

Type testing under simulated application conditions
MDC assists in the testing of high-level disinfectants for the automated reprocessing of flexible endoscopes, such as type testing of the efficacy of high-level disinfectants under use conditions in endoscope washer-disinfectors.
Biocompatibility
MDC assists in the preparation of a risk-based test concept according to ISO 10993 for the biological evaluation of medical devices, testing for leachables and extractables, literature searches and toxicological-chemical evaluation of residues.





